Finding the right vaccine technology
November 5, 2020 8 min read
As the search for a COVID-19 vaccine is intensifying and the demand for a distribution channel of the potential future vaccine is approaching, we decided to map up the potential vaccine technology platforms available.
We will break down the basis of each technology to gain a brief understanding of the challenges and possibilities of each vaccine. We will also briefly state the nature of the COVID-19 virus, to better understand its challenges in relation to the four technologies available.
" In the race towards Covid-19 vaccines, partnerships between developers and manufacturers are now therefore being established at an unprecedented level, with huge demand for equipment and consumables to enable manufacturing as a consequence."
- Dr. Ulrica Sehlstedt, Arthur D. Little
The nature of the COVID-19 virus.
The corona virus SARS-CoV-2 consists of a single strip of ribonucleic acid (RNA) surrounded by a shell of proteins.
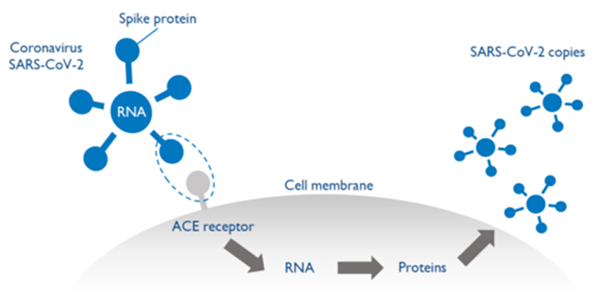
The shell contains protein spikes, which are critical for the virus to get inside cells and make copies of itself. SARS-CoV-2 uses its spike to bind to the ACE2 receptor, getting access into the cells. The virus’ RNA is then released into the cell. The cell reads the RNA and makes proteins that are then assembled into new copies of the virus.
The copies are released and go on to infect more cells. The vaccine typically activates the immune system’s T-helper cells, responsible for detecting the presence of a virus in the body. T-helper cells in turn instruct B-cells to develop antibodies that block the virus from getting inside the cells as well as activate T-killer cells to destroy infected cells.
There are four types of vaccine technologies.
Advancements in science and technology have given researchers new tools to try against the SARS-CoV-2 virus –there are four main platforms and common to all types of vaccines is their aim of teaching the body’s immune system to recognize and block viruses.
- Nucleic acid vaccines are based on RNA or DNA, generating spike proteins
- Viral-vectored vaccines use a different virus which has been engineered to be harmless to transport a SARS-CoV-2 virus gene into the cell
- Traditional subunit vaccines work by delivering viral proteins into the cell
- Another traditional approach is to use a weakened or deactivated form of the virus to trigger the immune system
Common to all type of vaccines is their aim of teaching the body’s immune system to recognize and block a certain virus
Vaccine technology 1: Vaccines using nucleic acid
Vaccines based on nucleic acids (DNA and RNA) are new and no such vaccine has yet been approved for use outside of medical research.
There are two main types of nucleic acid vaccines: DNA vaccines and RNA vaccines
- Both types lead to spike proteins being created inside the cells which help teach the immune system to recognize the virus
- These vaccines have the advantage of being able to design and manufacture quickly, but they have never been approved for use outside of medical research as of yet
Moderna’s mRNA-based covid-19 candidate vaccine was the first ever to enter human clinical trials in the US.
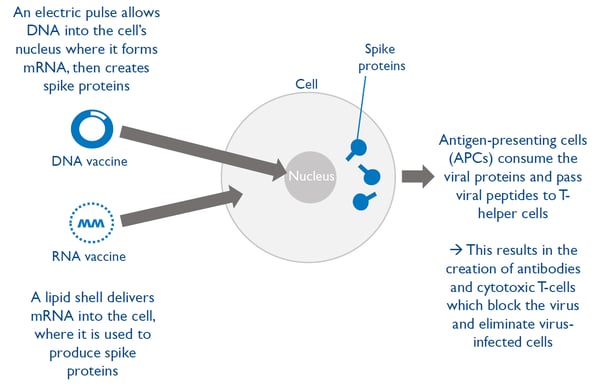
Pfizer and partner BioNTech are currently developing a RNA based vaccine that are currently in the phase of clinical trials. The intention, given that the trials generate positiv data, is to have the vaccine approved to hit the market by the 3rd week of November, says Albert Bourla, CEO at Pfizer.
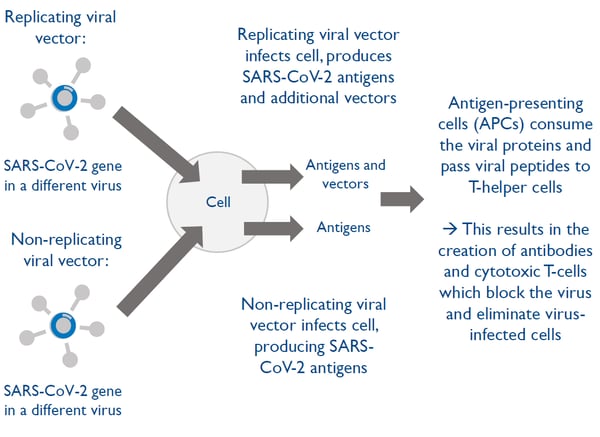
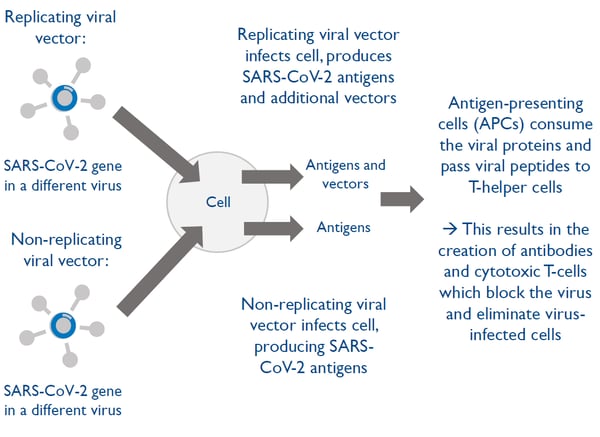
Vaccine technology 2: Viral-vectored vaccines
Adenoviral vector vaccines are one of the most promising vaccine types but so far, no human vaccine based on this technology has been approved.
Viral-vectored vaccines use a virus that has been engineered to be harmless in order to transport a gene from the virus into the cells
Viral-vectored vaccines can be designed and manufactured quickly however there is a risk of people developing immunity to the viral vector
There are two types of viral-vectored vaccines: replicating or non-replicating viral vectors.
Five out of the six viral-vector vaccines in clinical trials are based on adenovirus which is a technology that has been tested for 30 years but is used in only one commercial vaccine today: a rabies vaccine used to immunize wild animals.
So far, no human adenoviral vector vaccine has been approved.
Vaccine technology 3: Subunit vaccines
Traditional subunit vaccines deliver virus-like particles or protein subunits of the virus into the cells in order to trigger the immune system.
Subunit vaccines work by delivering viral proteins consisting of virus-like particles or protein subunits, into the cells.
For covid-19 candidate vaccines, protein subunits consisting of the virus’ spike proteins are used to trigger the body’s immune system.
The technologies used to manufacture subunit vaccines vary but a common technique is to use insect cells or yeast.
The technology has been used for many years and there are several examples of approved vaccines based on this platform.
One example is the hepatitis B vaccine which relies on a viral protein created by genetically engineered yeast.
Vaccine technology 4: Weakened or inactivated virus vaccines
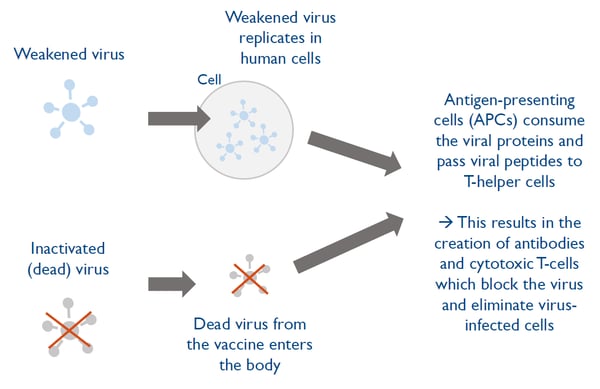
Vaccines based on a weakened or inactivated form of the virus constitute the most traditional approach but typically take long to manufacture.
Weakened or inactivated virus vaccines constitute the most traditional approach to vaccine development.
In weakened virus vaccines, the virus is weakened so that it does not cause any disease but still triggers the immune system’s defense mechanisms contain dead virus, incapable of infecting people but still able to instruct the immune system how to develop a defensive reaction against an infection.
Weakened and inactivated virus vaccines typically take longer to designand manufacture compared to newer technology approaches.
Reference: ADL report "mapping of covid-19 vaccines" commissioned by Metenova AB. September 4, 2020.
With a long experience in the engineering of magnetic mixers, and comprehensive knowledge of hands-on application – we keep challenging the limits of mixing technologies.





